Managing Drug Concepts
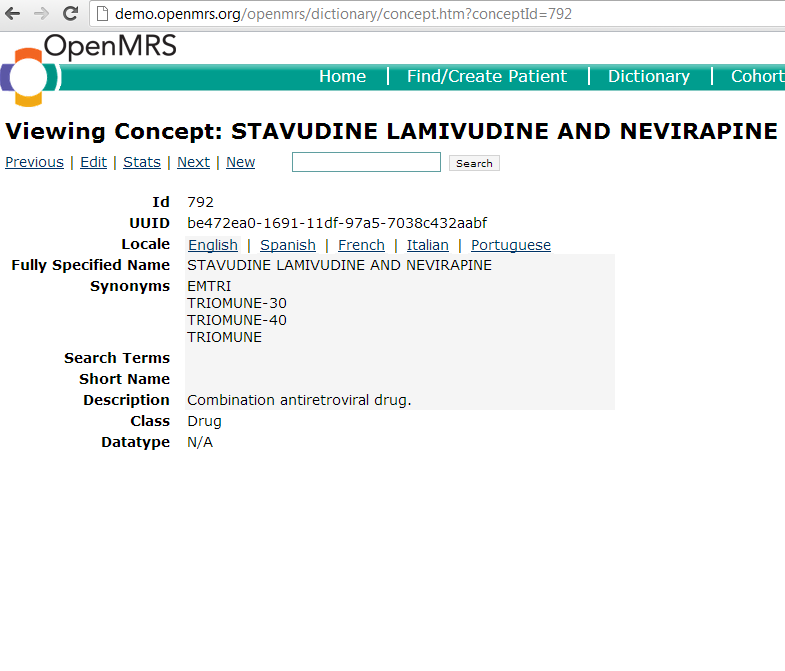
A drug concept is any concept with a "drug" class. The "drug" class should only be assigned to concepts that are drugs/medications. It is appropriate to create a drug concept anytime an implementation needs to store data about a particular medication. One should be sure to include all possible synonyms when creating the concept, as drugs oftentimes have many different names by which they may be referred. Once created, that this concept will reside in the concept table and can be used later to create different formularies if needed.
Here is an example of a drug concept:
Drug Formularies
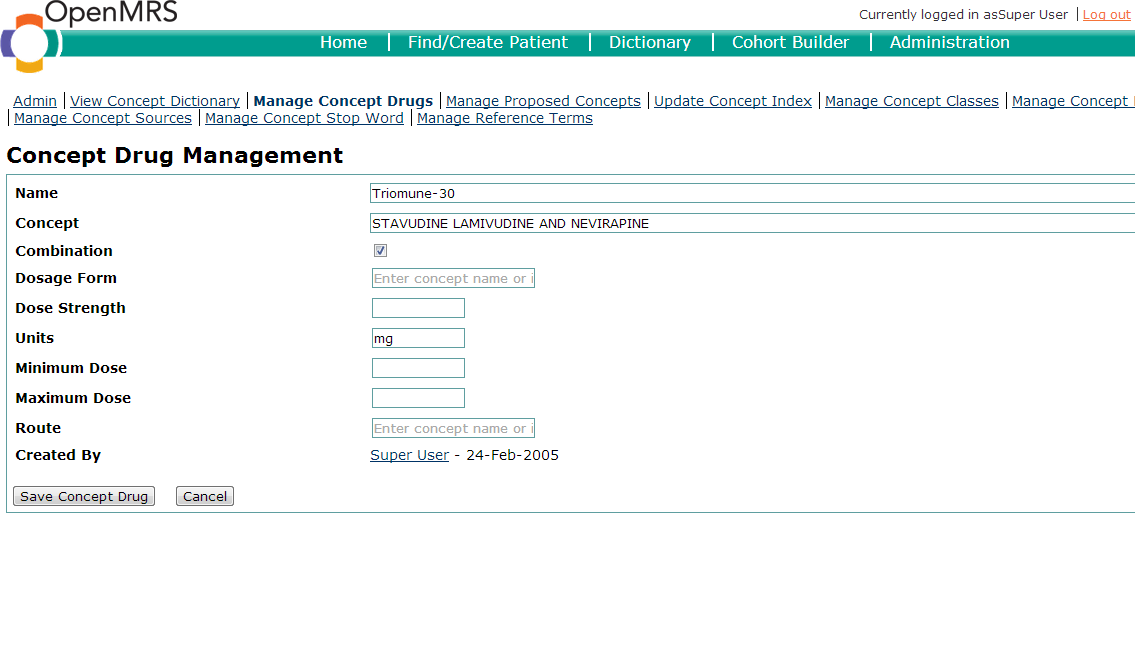
Drugs oftentimes have many different formulations. In OpenMRS, drugs and formulations have a 1:n relationship. In other words, each drug concept may have an unlimited number of drug formulations. Drug formulations may be created using the "Manage Concept Drug" page of the user-interface. When creating a drug formulation, you specify the drug concept to which the formulation is linked.
The following items should be defined in the creation of a drug formulary:
- Name - This is what the concept drug name should be. It is wise to use standard conventions for your implementation.
- Concept - This is the Concept ID which the concept drug is mapped to.
- Combination - Tick this box if the drug is a combination drug.
- Dosage Form - The preparation of the medication. Common forms include: Powder, syrup, tablet, capsule, solution,oral suspension, etc.
- Strength - The strength of the formulary. For example, an ISONIAZID 100 mg TAB would have a strength of 100 mg.
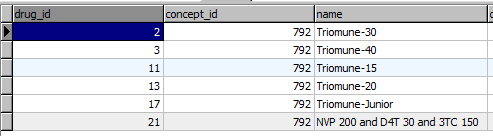
Each each drug formulation shares a concept_id with it's drug concept, but has a unique drug_id. The formulations are all stored in the drug table.
Best practices
Concept names for drugs should represent generic drug names (ideally mapped to RXNORM and SNOMED).
What are naming conventions for drug formularies used by different sites?
Concept | PIH | AMPATH |
|---|---|---|
Aspirin | Drug: Aspirin 200 mg tablet | ASPIRIN 200 mg TAB |
Ciprofloxacin | CPX (Ciprofloxacin 250 mg tablet) | CIPROFLOXACIN 500 mg TAB (CPX) |
Amoxicillin | Amoxicillin (125 mg/5 ml) |
|
Name dose formhttp://wiki.ampath.or.ke/pages/viewpage.action?pageId=36569685
Naming of Combination drugs
Split combination drug components by slash '/' , and put them in alphabetical order which is standard methodology used by RxNORM. AMPATH uses the full name of the drug since there are multiple shortname (ie. AZT and ZDV).
For example:
- Lamivudine 150 MG / Nevirapine 200 MG / Zidovudine 300 MG Oral Tablet
- 3TC 150mg / AZT 300 mg / NVP 200mg
Include the route in the drug name (ie. oral, IV, IM, topical,inhalation drug, etc). In the example of ISONIAZID 100 mg TAB, the drug route would be oral, so it should be Isoniqzid 100 mg tablet (oral).